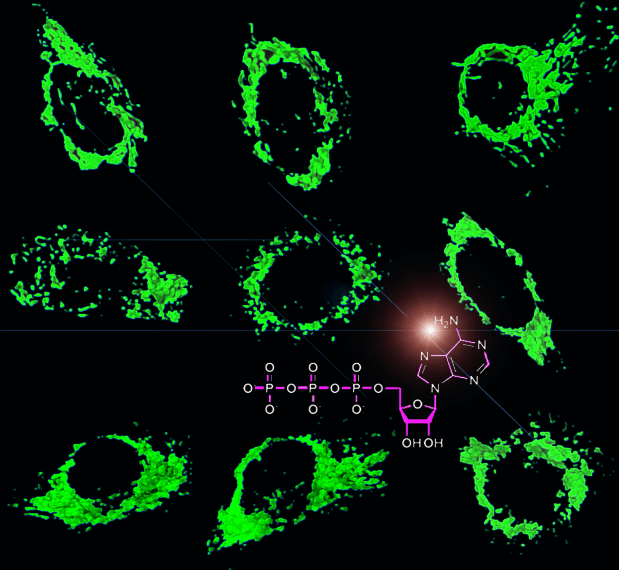
Mitochondria are central organelles involved in energy transduction to produce adenosine triphosphate (ATP) as well as energy signaling and cellular adaptations to various types of metabolic stress. The regulation of energy metabolism is multisite and includes:
- the direct modulation of respiratory chain kinetic parameters,
- modulation of OXPHOS intrinsic efficiency by changes in the basal proton conductance or the induced proton conductance,
- possible changes in the morphological state of the mitochondrial compartment,
- modulation of mitochondrial biogenesis and degradation, and
- in situ regulation of mitochondrial heterogeneity by the cellular and the mitochondrial microenvironment.
Therefore, mitochondrial physiology research is central to the study of energy metabolism, and the modulation of mitochondrial function is also a strategy of choice to restore energy metabolism homeostasis in a large array of diseases. Several inhibitors of the respiratory chain have been discovered so far, and used in a broad range of applications such as insecticide, piscicide, and pesticide.
However, only few activators have been discovered and few compounds are active on the mitochondrial membrane, organelle biogenesis and the F1F0-ATPsynthase activity. Mitochondrial biology includes a large number of specific features, as heteroplasmy, organelle dynamics, and the existence of a biochemical threshold effect which must be considered for the investigation of energy metabolism alterations and for the development of mitochondrial drugs.
In addition to their biological function as the cell powerhouse, mitochondria are now also considered as signaling platforms that control pathways involved in cell fate (the intrinsic pathway of apoptosis) and the immune response to pathogens and cell stress. These highly dynamic organelles host numerous innate immune signaling regulators, of which some are directly linked to the OXPHOS capacity and its control of oxidative stress. Therefore, mitochondrial bioenergetics is tightly connected to innate immunity and the extent of the immune response (Lartigue L, Faustin B; Int J Biochem Cell Biol. 2013 Sep;45(9):2052-6). Importantly, mitochondrial oxidative metabolism in T cells is instrumental to acquire their adaptive effector immune functions. Therefore, they emerge as promising therapeutic targets to improve T-cell mediated antitumor immune response, impair inflammation, or inhibit autoimmune processes (Anaplerotic metabolism of alloreactive T cells provides a metabolic approach to treat graft-versus-host disease. J Pharmacol Exp Ther. 2014 Nov;351(2):298-307).
- We investigate several aspects of mitochondrial function, structure, dynamics and composition
- We study mitochondrial signaling and interactions with cellular metabolism
- We can modulate mitochondrial structure and function
- We possess cell models with various types of mitochondrial injuries (toxic and genetic)
- We study mitochondria ex situ, in situ and in vivo







 Follow us on Linkedin!
Follow us on Linkedin!