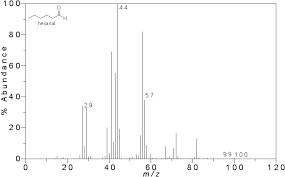
For the fourth in the analytical chemistry series, we turn to mass spectrometry. So far, we’ve looked at how chemists can determine the presence of particular bonds present in molecules with infrared spectroscopy, and how they can gain information about the structure of molecules using hydrogen and carbon nuclear magnetic resonance. Mass spectrometry allows us to measure the masses of atoms and molecules, and also obtain information about their chemical structure.
Before we talk about interpreting spectra, let’s discuss how they are generated in the first place. First, we need to generate ions from our sample. A small amount of sample is injected into the mass spectrometer, then it is ionised using an electron gun. The beam of electrons the electron gun generates knock electrons out of molecules of the sample, creating positive ions. In some cases, the positive ions generated can fragment further, producing a mixture of smaller positive ions and neutral molecule fragments. Most of the ions generated have a single positive charge, though some can have more.
Once the sample has been ionised, it is accelerated through an electric field before being passed through a magnetic field generated by an electromagnet. The positively charged ions are deflected as they pass through this field, with the magnitude of the deflection dependent on the mass/charge ratio of the ion. Lighter ions are deflected more, whilst heavier ions are deflected less. Ions with a larger charge are also deflected more. The mass spectrum is run under a vacuum to prevent the presence of anything other than the ions being tested.







 Follow us on Linkedin!
Follow us on Linkedin!
You must be logged in to post a comment.