Dr. Simran Sethi
Sr. Director, Consumer Research
Cliantha Research Limited, Sigma I Corporates, Sindhu Bhawan Road, Bodakdev, Ahmedabad – 380054. Gujarat, India.
E-mail: ssethi@cliantha.com Website: www.cliantha.com
Environmental pollution is a major external aggressors impacting skin health in modern urban populations. Persistent exposure to airborne pollutants and particulate matter has been linked to impaired skin barrier function, altered surface pH, increased inflammation, dullness, uneven tone, excessive buildup of impurities, and visible skin imperfections. With rising consumer awareness for anti‑pollution skincare solutions, robust clinical methodologies are essential to scientifically demonstrate protective and purifying effects under real‑world pollution conditions.
This case study highlights Cliantha Research’s capability in designing and executing comprehensive anti‑pollutionclinical studies, using an integrated assessment model that combines dermatological evaluations, advanced instrumental measurements, and structured subjective assessments. Cliantha enables cosmetic and dermocosmetic brands to secure robust, defensible, real‑world anti‑pollution claims that withstand regulatory scrutiny and resonate with consumers.
Study Overview
A single‑center, open‑label, single‑arm clinical study was conducted over a 28‑day period in healthy adult female volunteers aged 18–45 years. Participants were selected based on prolonged residence or occupational exposure in high‑air‑pollution environments (AQI >100) and the presence of pollution‑associated skin concerns, including blackheads, visible redness, uneven tone, blemishes, and rough skin texture.
Clinical assessments were conducted at predefined intervals throughout the study duration to monitor both immediate and progressive changes in skin condition.
Designed to Turn Pollution Stress into Market Advantage
Cliantha Research is a full-service Clinical Research Organization (CRO) committed to delivering high-quality, scientifically robust solutions. Cliantha’s anti‑pollution study designs are built to answer one core commercial question: Does your product perform under real environmental stress—and can you confidently claim it?
To answer this, Cliantha designs targeted, claim‑driven studies, allowing meaningful measurement of cleansing, protection, and skin quality improvement.
The result: data that reflects real life, not laboratory theory.
Three‑Fold Anti‑Pollution Assessment Strategy
Cliantha integrates a three‑pillar clinical framework designed to secure both scientific credibility and commercial impact.
1. Dermatologist‑Led Clinical Evaluation
Dermatologist‑led clinical assessments provided expert visual and tactile evaluation of pollution‑related skin parameters. Qualified dermatologists performed standardized grading of:
- Deep cleansing performance, determined through changes in visible blackhead count
- Evenness of skin tone, assessed using a validated clinical grading scale
- Extent of visible skin redness, evaluated through intensity‑based scoring
- Skin glow and radiance, assessed using a validated clinical grading scale
These evaluations enabled clinically meaningful interpretation of skin responses related to pollutant accumulation, surface congestion, and inflammation, ensuring strong medical relevance of study outcomes.
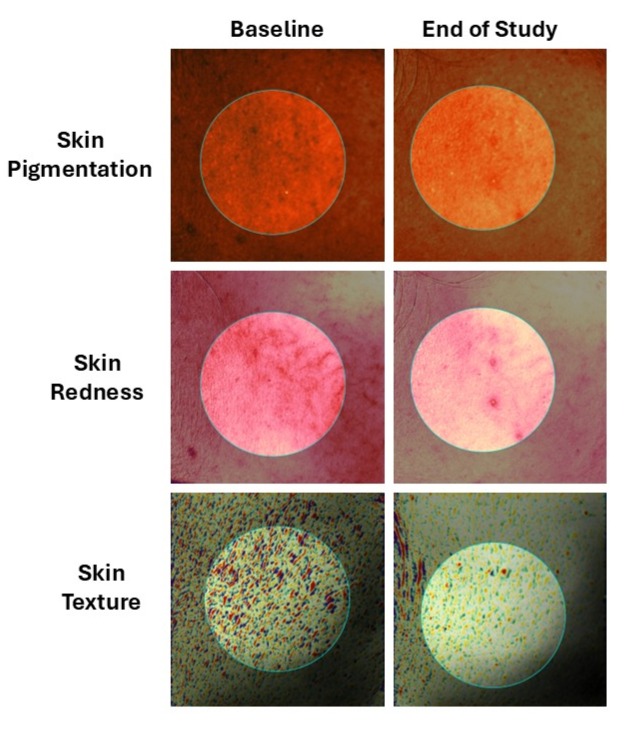
2. Advanced Instrumental Proof
Objective quantification of skin properties was achieved using non‑invasive, validated instrumental technologies to capture functional and structural changes induced by environmental pollution:
- Skin glow and radiance, measured using a reflectance‑based gloss evaluation system
- Skin barrier function, assessed through Transepidermal water loss measurements
- Surface pH balance, determined using a precision skin pH measurement device
- Skin hydration levels, quantified via capacitance‑based assessment technology
- Skin blemishes, redness parameters, and surface roughness, analyzed using high‑resolution three‑dimensional imaging
Together, these instrumental parameters supported precise, reproducible measurement of anti‑pollution efficacy beyond subjective observation.
3. Subjective & Consumer‑Reported Outcomes
To complement clinical and instrumental data, structured subject self‑assessment tools were employed to capture user perception. Participants evaluated attributes such as skin cleanliness, comfort, softness, soothing effect, gentleness, perceived brightness, and overall skin feel at scheduled visits. This dimension ensured correlation between measured biological effects and perceived anti‑pollution benefits from the end‑user perspective.
Data Handling & Safety Evaluation
Study data was analyzed using inferential statistical methods, with predefined significance criteria. Safety assessment was conducted throughout the study via investigator observation and spontaneous reporting of any adverse events, ensuring tolerability and regulatory compliance.
Conclusion
This case study demonstrates Cliantha’s end‑to‑end expertise in anti‑pollution clinical testing, supported by a three‑pillar evaluation framework integrating dermatologist insight, advanced instrumental analysis, and validated subjective feedback. Such a multidimensional approach provides robust, scientifically credible evidence to support anti‑pollution performance claims and reflects best practices in modern cosmetic clinical research conducted under real‑world urban pollution conditions.
CLIANTHA – A Strategic Clinical Partner for Anti‑Pollution Claims
With expertise spanning Clinical Trials (Phase I–IV), Bioavailability/Bioequivalence studies, Dermatology research, and Consumer Healthcare, Cliantha conducts high-quality clinical trials across pharmaceuticals, biologics, cosmetics, personal care, and nutraceuticals. At Cliantha, every trial, every study, and every breakthroughs are a step toward enhancing consumer safety, efficacy, and trust.
- State-of-the-art clinical facility – Equipped with advanced bio-instruments and experienced technical work force.
- Volunteer Database – Having categorization of population with continuous pollution exposure.
- Claim‑Driven Study Design – Protocols aligned with marketing objectives from day one.
- Regulatory‑Ready Data – Statistically analyzed, safety‑monitored, globally usable study outputs.
- Proven Cosmetic & Dermocosmetic Experience -Trusted by global brands to support high‑value efficacy claims.
Cliantha doesn’t just test products. We help brands prove performance where it matters most.
Contact
Dr. Simran Sethi
Sr. Director, Consumer Research
E-mail: ssethi@cliantha.com
Website: www.cliantha.com

To know more about Cliantha Research expertise: https://www.skinobs.com/c/labo.php?id=153




