The skin microbiome has evolved from scientific curiosity to commercial imperative. Today’s consumers actively seek products that support skin health through microbiome balance, driving brands to substantiate increasingly sophisticated microbiome-related claims. Yet behind these marketing messages lies a significant technical challenge: accurately profiling the skin microbiome remains frustratingly complex.
For years, cosmetic researchers have relied on DNA sequencing technologies—primarily 16S rRNA sequencing and shotgun whole genome sequencing (WGS) – despite their well-documented limitations. These methods present significant obstacles including poor taxonomic resolution, prohibitive costs, host DNA interference, and extended turnaround times that can delay product launches by months. As the industry races to develop microbiome-optimized formulations, these technical constraints have become a critical bottleneck in the innovation pipeline.
The Technical Reality of Skin Samples
Skin samples are inherently challenging. They typically contain low microbial biomass alongside overwhelming quantities of host DNA, often exceeding 90% of total extracted DNA. This creates significant problems for sequencing-based approaches. Additionally, species-specific effects matter enormously: organisms within the same genus can have vastly different impacts, with some being protective while others prove pathogenic. Without species-level resolution, researchers cannot accurately understand how products interact with the skin microbiome.
16S rRNA sequencing, long considered the standard approach, provides only genus-level taxonomic resolution. Shotgun WGS offers superior resolution but requires expensive host DNA depletion procedures, deep sequencing runs, and complex bioinformatics analysis driving both costs and timelines to levels that many projects cannot sustain.
Perhaps most problematic, both sequencing approaches report relative abundance rather than absolute quantification. This fundamental limitation can produce misleading results, as changes in one species artificially affect the apparent abundance of all others obscuring true biological effects.
A Quantitative Alternative: Precision Microbiome Profiling (PMP™)
Addressing these limitations, Bio-Me developed PMP™ (Precision Microbiome Profiling), a comprehensive qPCR-based approach specifically designed for in vivo skin microbiome research. Unlike sequencing methods, targeted qPCR is inherently unaffected by host DNA and delivers absolute quantification alongside species and subspecies-level resolution.
The PMP™ Skin Microbiome panel quantifies 52 key bacterial and fungal species in a single test, providing comprehensive coverage across diverse skin types and anatomical regions. This includes critical species for applications such as dandruff management (Malassezia restricta, Cutibacterium acnes, Staphylococcus epidermidis), odor control (Staphylococcus hominis, S. lugdunensis), and broader concerns including inflammation, barrier function, UV response, and aging. Key PMP™ features translate into multiple benefits:
- Quantitative
- Accurate and reproducible
- Fast turnaround time – in days
- Data is clear and actionable

Independent validation from researchers at MIT compared PMP™ against both 16S and shotgun WGS across 43 human leg skin samples. The study demonstrated that PMP™ accurately captured microbial diversity at levels comparable to shotgun sequencing, while 16S sequencing failed to detect low-abundance genera, presenting a falsely Cutibacterium-dominant profile that misrepresented true community structure (figure 1).
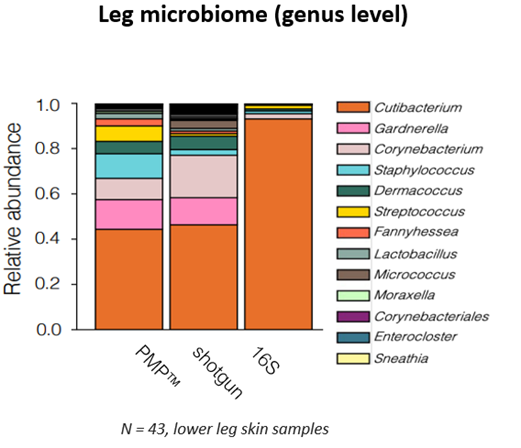
Practical Advantages for Product Development
The implications for cosmetic innovation are substantial. PMP™ delivers Quantitative, Reproducible results in days rather than weeks, at a fraction of sequencing costs. For R&D teams, this translates directly to
- Accelerated development timelines
- More confident formulation decisions, and
- Robust data for regulatory submissions and marketing claims.
Absolute quantification reveals true microbial load and treatment effects without the confounding factors inherent to relative abundance data. Species-level precision enables targeted product development based on specific microbial interactions rather than genus-level assumptions. Simple, interpretable data reports eliminate the need for specialized bioinformatics expertise, making results immediately actionable for product development teams.
Meeting Diverse Research Needs
Beyond standard skin applications, Bio-Me has expanded the PMP™ platform to address specialized cosmetic segments. An Intimate Care panel combines 52 skin and vaginal species for products targeting intimate health. A dedicated 52 target Vaginal Microbiome panel enables Community State Typing for comprehensive vaginal health assessment. For ingestible beauty applications, a Gut Microbiome panel profiles 108 species relevant to skin health “from within”.
The Path Forward
As microbiome science continues reshaping cosmetic innovation, the industry requires analytical tools that match the pace of discovery. By delivering species-level precision with quantitative accuracy in commercially relevant timeframes, targeted qPCR approaches like PMP™ are establishing a new standard for skin microbiome research transforming what was once a bottleneck into a competitive advantage.
Bio-Me AS, based in Oslo, Norway, provides comprehensive microbiome testing solutions to cosmetic companies, contract research organizations, and academic researchers worldwide.
For more information, visit www.bio-me.com






