Scientific • Clinical • Strategic perspective on turning microbiome data into decisions
The microbiome has shifted from an exciting research frontier to a strategic asset for innovation across human health, animal health, cosmetics, food, and environmental monitoring. Yet the field still faces a practical paradox: we can generate abundant sequencing data, but too many programs struggle to turn that data into conclusions that are robust, transferable, and decision-ready.
Microbiome signals are inherently contextual. Diet, medications, seasonality, geography, sampling conditions, and even the choice of extraction kit can alter the observed community. Low-biomass matrices are also vulnerable to background contamination, and analytical choices (filters, databases, statistical models) can change the apparent signature. If these sources of variation are not controlled, results may look compelling in a single dataset but fail to replicate across sites, time, or cohorts.
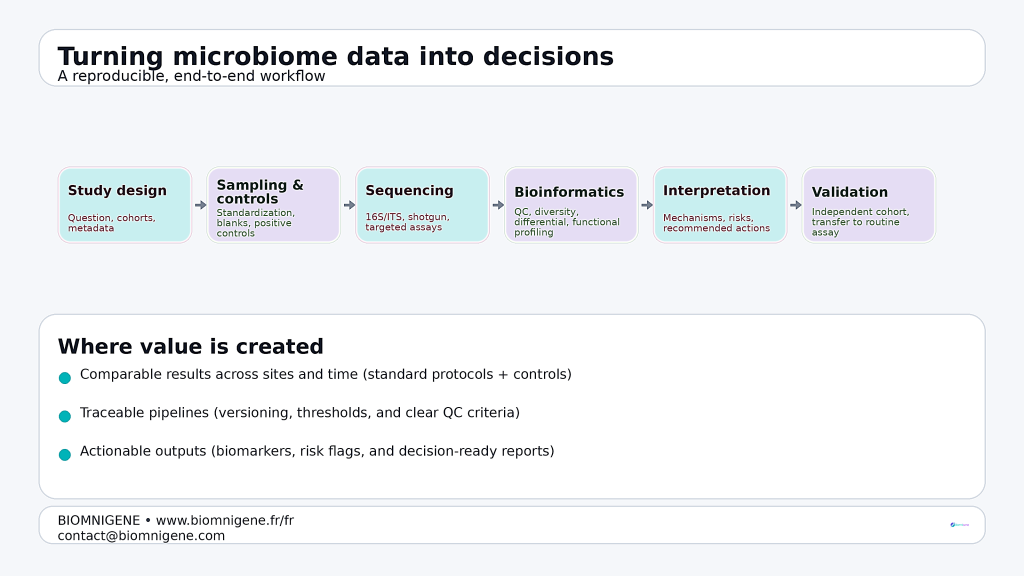
In practice, value is created less by sequencing alone and more by mastering the entire chain: study design, standardized sampling, rigorous controls, traceable bioinformatics, and interpretation that supports action. This is where microbiome work begins to resemble regulated diagnostics and industrial quality systems: reproducibility is not a nice-to-have; it is the foundation of credibility.
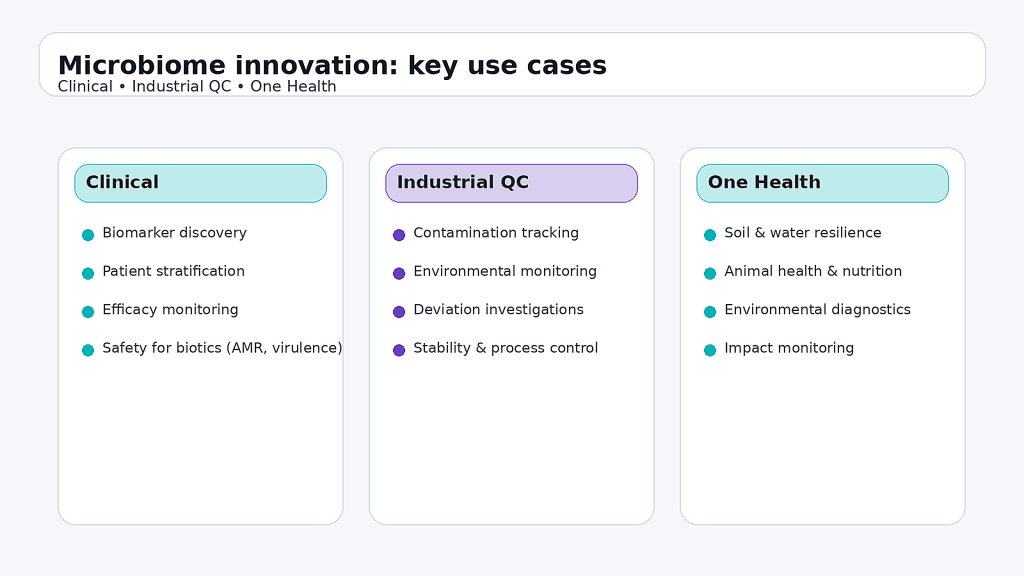
From a clinical perspective, a microbiome biomarker only matters if it changes a decision. That decision can be patient stratification, risk monitoring, response prediction, or safety assessment for biotics, including screening for antibiotic resistance and virulence-related risk. To reach that level, projects need an evidence pathway similar to clinical diagnostics: a clear intended use, predefined endpoints, high-quality metadata, and validation on independent cohorts. Technology should be selected based on the objective and maturity of evidence: 16S/ITS for scalable community-level shifts, shotgun metagenomics for higher resolution and functional potential, and targeted qPCR or dPCR for verification and routine monitoring. The end point is not a prettier plot, but a result that can be trusted when applied to new patients and new settings.
On the industrial side, microbiome analytics is rapidly becoming a quality and risk-management tool. In bioproduction and advanced manufacturing, it can support environmental monitoring, contamination tracking, and deviation investigations, helping teams interpret atypical events such as yield drops, instability, or unexpected signals with greater resolution than classical approaches alone. In dermocosmetics, microbiome readouts can strengthen mechanistic understanding and claims when paired with robust controls and appropriate statistics. In One Health programs, microbiome metrics can inform ecosystem monitoring across soils, water, animals, and humans, providing early indicators of stress, resilience, or perturbation.
Across these domains, five pillars consistently determine whether a microbiome project delivers high value:
1. Rigorous study design: define the question, minimize confounders, collect meaningful metadata, and plan for statistical power.
2. Standardized sampling and handling: control what happens before the lab (devices, preservatives, timing, temperature, shipping), because variability introduced here cannot be corrected downstream.
3. Fit-for-purpose controls: include extraction blanks and positive controls, and apply contamination-aware practices, especially for low-biomass samples.
4. Auditable analytics: run versioned, traceable pipelines with transparent thresholds, clearly stated reference databases, and well-reported QC metrics.
5. Decision-ready interpretation: translate results into what stakeholders can do next (what is supported, what remains uncertain, and what should be validated).
At BIOMNIGENE, we focus on microbiome programs rather than isolated experiments. That means end-to-end stewardship: protocols aligned with the objective, robust controls, transparent QC criteria, and reporting designed for decisions. Our broader vision is to enable microbiome work that is both industrial-grade and clinical-grade, and to accelerate transferability by integrating high-quality metadata and scalable analytics, including AI-driven approaches when relevant.
The next wave of microbiome innovation will be defined by reproducibility and utility: results that hold up beyond a single dataset, and insights that teams can trust and act on. Whether your goal is a clinical biomarker, a manufacturing risk signal, or a One Health monitoring program, the key question is not Can we sequence, but Can we trust the conclusion and use it.
Supporting visuals
Workflow diagram
Use-cases overview (clinical, industrial QC, One Health).
Contact
Mr Alexandre Douablin
contact@biomnigene.com